
Elite iQ
The Elite iQ is a modern, intelligent dual-wavelength aesthetic platform enabling practitioners to perform many of the most requested aesthetic treatments. Higher style. Higher power. Higher iQ.
Ready to find your beautiful? Get started by locating a Cynosure provider near you.
Find a providerInnovative technology. Best-in-class solutions. All to enhance your practice.

The Elite iQ is a modern, intelligent dual-wavelength aesthetic platform enabling practitioners to perform many of the most requested aesthetic treatments. Higher style. Higher power. Higher iQ.
One multi-purpose platform that provides a comprehensive range of popular treatments.

LIPOcel is a state-of-the-art Non-Invasive, Non-Surgical treatment for focal fat reduction and precision body contouring; treating the flanks, stomach, arms, knees and back of thighs. Impressive Results; 20 to 40% Reduction*

The newer, faster, stronger PicoSure Pro harnesses the power of picosecond technology to tackle pigmentary concerns, fine lines, texture, tattoo removal and more.

The world’s first RF microneedling system that combines monopolar and bipolar RF at 1 or 2MHz frequencies in a single device.
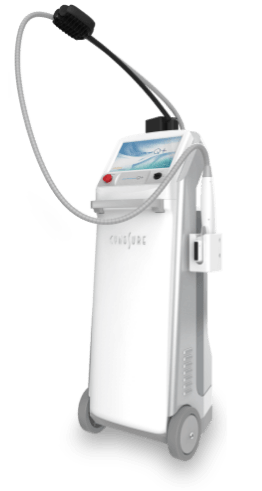
ULTRAcel Q+, a next generation HIFU technology which treats fine lines and wrinkles, tightens skin and removes stubborn fat cells.
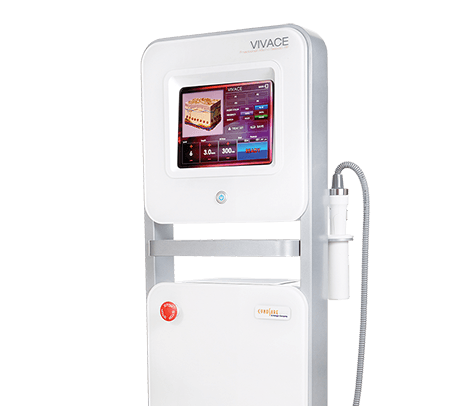
The newest generation of radiofrequency microneedling that is FDA cleared and TGA listed.

CynoGlow is a customisable, sequential treatment plan utilising the most advanced picosecond laser, Picosure Pro and radio frequency micro-needling technology, Potenza.

The Elite iQ is a modern, intelligent dual-wavelength aesthetic platform enabling practitioners to perform many of the most requested aesthetic treatments. Higher style. Higher power. Higher iQ.

A dual-wavelength system for hair removal, vascular and pigment treatments, and wrinkle reduction.
One multi-purpose platform that provides a comprehensive range of popular treatments.

LIPOcel is a state-of-the-art Non-Invasive, Non-Surgical treatment for focal fat reduction and precision body contouring; treating the flanks, stomach, arms, knees and back of thighs. Impressive Results; 20 to 40% Reduction*
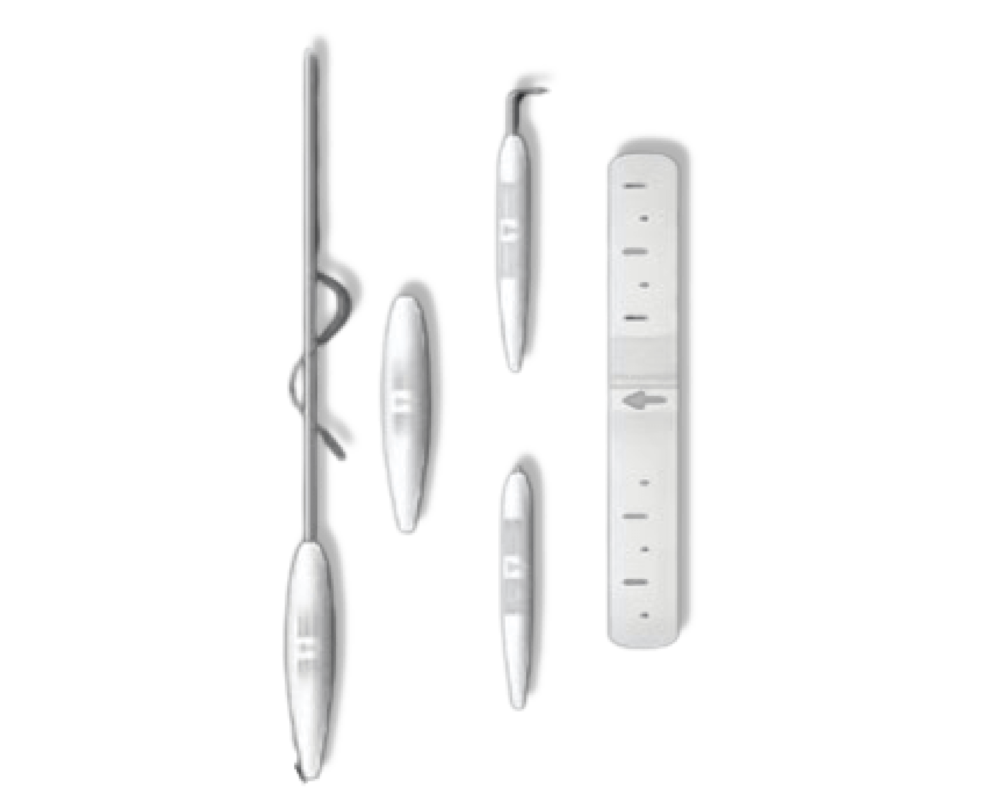
A comprehensive jawline contouring procedure that offers the convenience of minimally invasive technology with long-lasting results approaching those of full surgical intervention.

The newer, faster, stronger PicoSure Pro harnesses the power of picosecond technology to tackle pigmentary concerns, fine lines, texture, tattoo removal and more.

The World's No.1 and first picosecond aesthetic laser for skin revitalisation and pigmentary treatments.

The world’s first RF microneedling system that combines monopolar and bipolar RF at 1 or 2MHz frequencies in a single device.
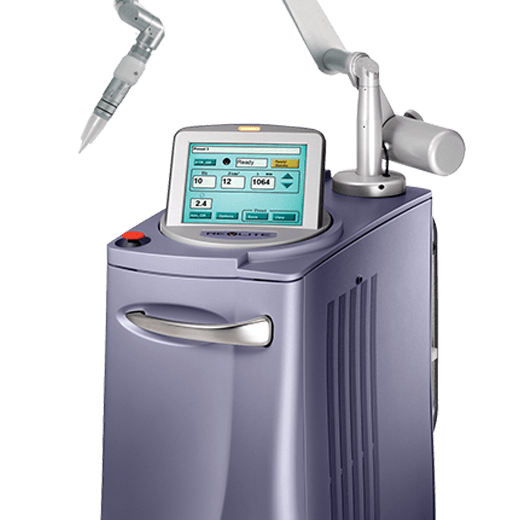
High-powered Q-Switched Nd: YAG laser for skin revitalization and pigmentary and tattoo treatments.

StimSure® is CE marked for muscular atrophy and can be used to strengthen and tighten the abdominal, gluteal and thigh muscles through methods of contraction and stimulation

ULTRAcel Q+, a next generation HIFU technology which treats fine lines and wrinkles, tightens skin and removes stubborn fat cells.

The newest generation of radiofrequency microneedling that is FDA cleared and TGA listed.

The ZWave Pro is a FDA cleared and TGA listed radial pulse technology that delivers acoustic shockwaves to reduce the appearance of cellulite.
Keep your practice on top with the latest best-in-class offerings along with dedicated support and turnkey solutions when you partner with Cynosure.